News
Health Alert: Pfizer Recalls More Blood Pressure Pills Over Cancer Risk
Pfizer has recalled more of its blood pressure medications over health risk associated with a potentially cancer-causing chemical impurity. The company has recalled some lots of it blood pressure pill Accuretic and a few of generic drugs used to treat hypertension.
Every Day Health reported today (March 24, 2022) that Pfizer has voluntarily recalled some lots of Accuretic pills due to the presence of elevated levels of nitrosamine, a chemical that can increase the risk of cancer.
Pfizer said it is not aware of any adverse events associated with nitrosamine impurities in the recalled lots of Accuretic tablets.
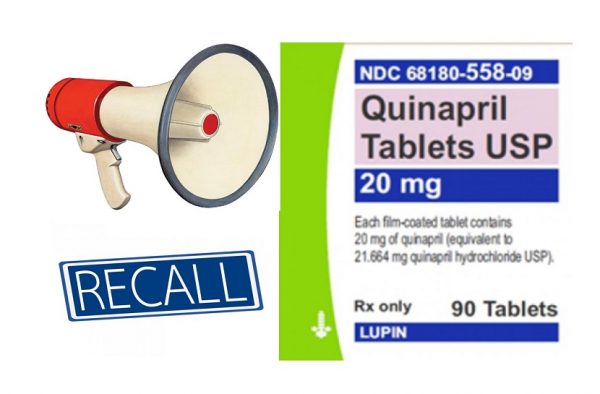
In addition to Accuretic, Pfizer has also recalled select lots of Quinapril and Quinapril HCl tablets. Cleveland.com reported that one lot of Quinapril hydrochlorothiazide tablets and four lots of Quinapril HCl/ hydrochlorothiazide tablets have been recalled. These generic drugs are distributed by Greenstone.
The medications were distributed nationwide to wholesalers and distributors in the U.S. and Puerto Rico from November 2019 to March 2022.
Pfizer says it has asked the distributor to stop distribution of these recalled drugs.
Nitrosamine impurities are said to exist in some drugs because of the way they are manufactured, stored, and/or packaged. The acceptable intake limit set by the FDA of 26.5 nanograms of nitrosamine per day (ng/day).
Earlier this month, Pfizer in Canada recalled all lots of Inderal-LA capsules over similar concerns of nitrosamine impurity.
For information on how to return the recalled pills and get a refund, call 888-843-0247 during weekday business hours.